The mRNA Revolution and the Future of Canadian Medicine
We often think of medical breakthroughs as rare “lightning in a bottle” moments achieved by a few ‘great minds’ with a single moonshot—like the 1922 discovery of insulin by Canadian scientists Frederick Banting and Charles Best, or penicillin in 1928 by Scottish physician Alexander Fleming.
But such revolutionary discoveries emerge after many phases of research conducted by teams of talented scientists over decades have laid the groundwork for ground-breaking—and life-saving—medicine.
This is the case with the development of Messenger RNA (mRNA) medical technology, which began in 1961 with scientists testing its effectiveness against various diseases, and culminated in a world-changing breakthrough in 2020 through the arrival of the first COVID-19 vaccine.
Perhaps it’s because this kind of revolutionary scientific discovery occurred amid a frantic race for life-saving treatment during an unprecedented global pandemic that the development of safe and effective vaccines is commonly viewed as the pinnacle of mRNA innovation. But the medical community has only just begun harnessing this tiny particle’s potential and is already embarking on new and novel ways to advance its technology.
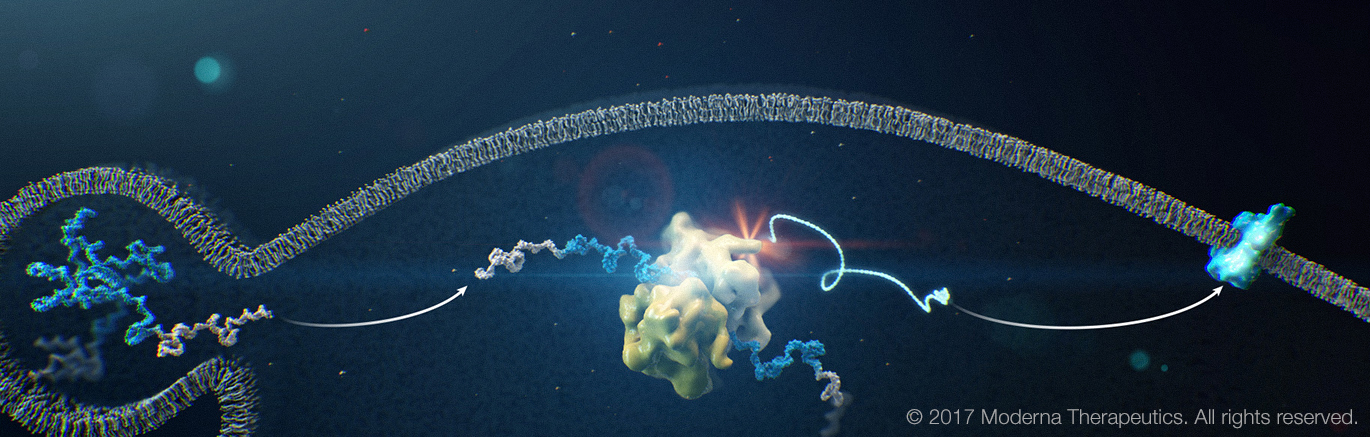
mRNA—the key to understanding fundamental systems that govern our bodies—is a single-stranded molecule made from a DNA template that teaches the body how to make proteins.
By introducing a small fragment of the molecule through an injection, an mRNA vaccine provides instructions for the body to make a specific protein found on a virus’s surface and works with the immune system to recognize and fight future exposure.
The mRNA then breaks down quickly, leaving the recipient’s DNA completely unaltered even as the body becomes naturally immune to a specific disease.
In harnessing mRNA’s innate biological processes, scientists have created a powerful tool that could enable the human immune system to conquer diseases that have plagued humanity for centuries.
As the world witnessed the race to develop, test and distribute a viable COVID-19 vaccine on a global scale, the mRNA vaccine developed by Cambridge, Mass.-based biotechnology company Moderna emerged as a promising frontrunner for a safe and effective bulwark against the virus’ spread.
Moderna has been pioneering mRNA technology to create vaccines and therapies for a range of common viruses and rare diseases, such as cancer, HIV, respiratory, and metabolic disorders, for more than a decade. By 2020, Moderna had advanced nine mRNA vaccine candidates for testing against various infectious diseases but had yet to bring a commercial product to market.
“Moderna was very much starting from scratch as a commercial organization in the pandemic—the Canadian team only had about five people during the peak—but our [mRNA] platform enabled us to develop, test and bring a new vaccine to market with significant speed and scale.” Says Dr. Shehzad Iqbal, Country Medical Director and one of Moderna Canada’s first employees. “As a company we punched above our weight to generate a significant impact during a time of great uncertainty.”
When the global pandemic was declared in March 2020, Moderna quickly narrowed their focus and created a prototype vaccine within days of COVID-19’s genome sequence becoming available online.
In under ten weeks, Moderna collaborated with the U.S. National Institute of Allergy and Infectious Diseases (NIAID) to conduct studies and launch trials of their new mRNA vaccine. Moderna’s candidate vaccine demonstrated a 94 per cent efficacy rate at preventing COVID-19 symptoms in adults during initial clinical trials.
mRNA vaccines for COVID-19 were granted use by Health Canada under Interim Order in December 2020, and Canada became the first country to fully approve Moderna’s vaccine in 2021. According to Health Canada, of the more than 98 million vaccine doses administered to Canadians from December 2020 to date, over 96 million are mRNA vaccines, with Moderna’s accounting for nearly 30 million.
As Moderna’s first commercial product, their COVID-19 vaccine represented a transformative milestone for the young biotechnology company. More importantly, mRNA’s now proven potential has signaled a dramatic shift in global medicine.
“mRNA technology is versatile and customizable, enabling targeted treatment with faster response times to a variety of diseases,” explains Iqbal. “With ongoing advancements, we can imagine a future where mRNA therapies become the standard of care, offering focused and personalized treatments for a wide range of medical conditions.”
With mRNA technology playing a transformative role in the future of modern medicine, Moderna is leading the way in its development with Canada serving as a trusted partner in research and development.
In August 2021, Moderna announced a 10-year partnership with the federal government to support their Biomanufacturing and Life Sciences Strategy in part through the construction of an mRNA vaccine facility in Laval, Que. Once completed in 2025, subject to planning and regulatory approvals, the facility will produce a domestic supply of respiratory vaccines and help rebuild Canada’s domestic biomanufacturing sector.
Combined with Moderna’s other collaborations coast-to-coast-to-coast, this long-term partnership is expected to further bolster the expertise of Canada’s life sciences ecosystem and position the country as a global centre of excellence in mRNA technology.
As Canadians navigate the possibilities and implications of mRNA for global health care, seeking accurate information from reliable sources, such as the World Health Organization (WHO) and Health Canada, and engaging in informed discussions through critical inquiry is essential.
“The potential to transform health care through mRNA is immense,” says Iqbal. “We’ve only begun to scratch the surface of what this technology can achieve.”
To stay up-to-date with Moderna’s latest developments and be a part of the conversation shaping the future of health care, join their LinkedIn Group and visit modernatx.com.
